 
this type of band is also obtained when white light is passed through diluted solution of blood or chlorophyll or through certain solutions of organic and inorganic compounds. 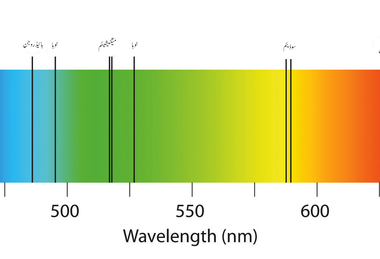 
When the white light is passed through the iodine vapour, dark bands on continuous bright background is obtained. Similarly, if the light from the carbon arc is made to pass through sodium vapour, a continuous spectrum of carbon arc with two dark lines in the yellow region of sodium vapour is obtained. This causes an absorption spectra, as shown below. When light from the incandescent lamp is passed through cold gas (medium), the spectrum obtained through the dispersion due to prism is line absorption spectrum (figure). Absorption spectra When photons of light pass through a gas, the photons with the same energy as the energy gaps in the atoms can be absorbed. this is an example of continuous absorption spectrum. A spectrum is simply a chart or a graph that shows the intensity of light being emitted over a range of energies. Therefore, absorption of light during electronic excitation causes there to be absent frequencies/wavelengths of light in the otherwise continuous spectrum. For instance, when we pass white light is passed through a blue glass plate, it absorbs everything except blue. When the light is passed through a medium, it is dispersed by the prism, is obtained continuous absorption spectrum. An emissions spectrum looks like a set of colored lines on a black background as opposed to an absorption spectrum which looks like black lines on a colored. Absorption spectrum is classified into three types: the inverse of a visible absorption spectrum. It is the characteristic of absorbing substance. When light is allowed to pass through a medium or an absorbing substance then the spectrum obtained is known as absorption spectrum. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
